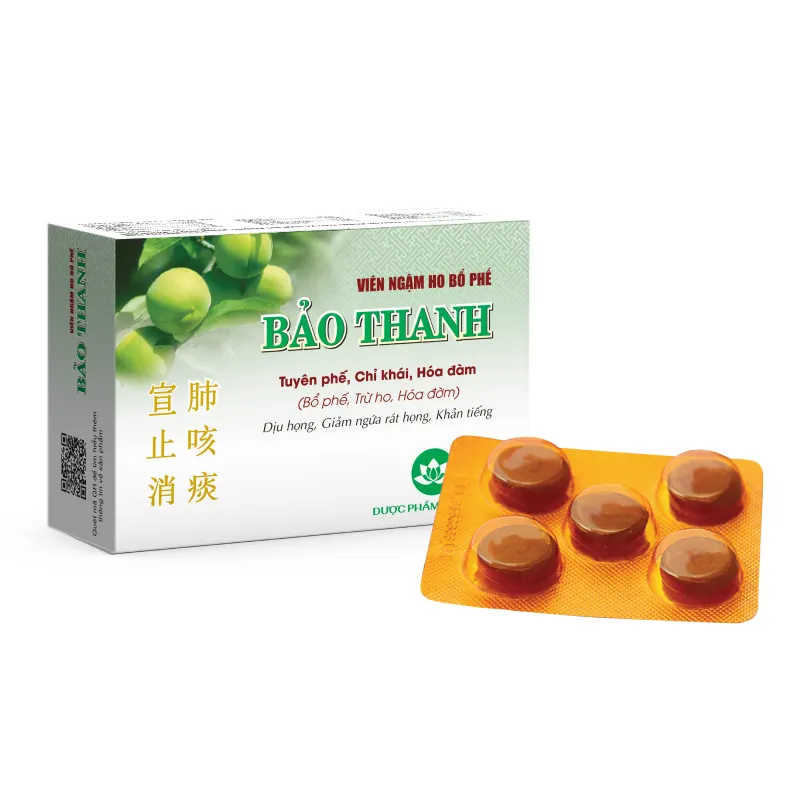
Bao Thanh Cough Suppressant Lozenges
(0 Ratings)
Share
Brand
Bao Thanh
Grade / Type / Style
Box of 20 pills, Box of 100 pills
Expiration Date
24 months from the date of manufacture
Food form / shape
Tablet form
Preservation method
Dry place, temperature not exceeding 30°C, Avoid sunlight
Flavor
Sweet, slightly bitter
Not applicable to people
Do not use lozenges for children under 3 years old, Children with a history of epilepsy or seizures
Receive processing
No
Minimum Order Quantity (MOQ)
1,000 box
Provide samples
No
Product Condition
100% New
Export Port
Cat Lai
Supply Quantity
2000
Main Export Market
Taiwan, Hong Kong, Malaysia
Product Certificate
ISO 9001 - International Quality Management System
Packaging
Packaging type
box
Packaging Detail
Box of 4 blisters x 5 lozenges; Box of 20 blisters x 5 lozenges
Packaging dimensions (length x width x height)
12 cm x 5 cm x 5 cm
Packing weight
500 g
B2B Sales Policy
As per sales agreement
As per sales agreement
As per sales agreement
Detailed description
Bao Thanh supports lung health, relieves coughs, and thins phlegm.
Improves lung health and helps reduce recurrence. Good for the whole family!
Formula Composition: Per lozenge
Platycodon grandiflorum - 0.2g
Mukdenia rossii - 0.5g
Anemarrhena asphodeloides - 0.1g
Poria cocos - 0.1g
Citrus reticulata - 0.1g
Blumea balsamifera - 0.4g
Pinellia ternata - 0.1g
Schisandra chinensis - 0.05g
Trichosanthes kirilowii - 0.2g
Polygala tenuifolia - 0.1g
Prunus persica - 0.2g
Zingiber officinale - 0.1g
Salted apricot - 0.5g
Ophiopogon japonicus - 0.1g
Asparagus cochinchinensis - 0.05g
Glycyrrhiza uralensis - 0.1g
Peppermint oil - 1.1mg
Honey - 1.0g
Excipients: White sugar, malt sugar, benzoic acid, 96% ethanol, purified water to make 1 lozenge.
Dosage form: Lozenge, yellowish-brown color, cool scent, sweet taste, slightly bitter.
Effects
Expectorant, cough suppressant, phlegm-thinning (Supports lung health, relieves coughs, and thins phlegm)
Indications
– Coughs due to colds, chilling, wind-cough, dry cough, productive cough, weather-related allergy coughs.
– Individuals with lung yin deficiency causing persistent cough, dry mouth and throat, throat itching, burning sensation, hoarseness.
– Supports the treatment of pneumonia, pharyngitis, and bronchitis.
Dosage and administration:
● Children aged 3-10 years: 1 lozenge per time, 3-4 times a day.
● Children over 10 years: 1 lozenge per time, 5-6 times a day.
● Adults: 1 lozenge per time, 6-8 times a day.
Suck until dissolved or chew before swallowing.
Contraindications: Do not use lozenges for children under 3 years of age (due to choking risk), children with a history of epilepsy or seizures during high fever. Do not use this medicine for individuals sensitive to any component of the medicine.
Warnings and precautions when using the medicine: Use with caution in pregnant women.
Use of medicine for pregnant women and breastfeeding: There is no data on the effects of the drug components on pregnant and breastfeeding women. Use caution when using for pregnant women; only consider if the benefits outweigh the risks.
Drug interactions, incompatibilities: No incompatibility has been reported. Do not mix this medicine with other medicines.
Undesirable effects: No reports of undesirable effects from the medicine have been recorded. Immediately report any undesirable effects encountered when using the medicine to your doctor or pharmacist.
Effects of the medicine on driving ability and operating machinery: There is no evidence of the medicine’s effect on driving ability and operating machinery.
Overdose and management: There is no data on overdose use, do not use beyond the prescribed dosage. Actively monitor for timely treatment.
Note: Do not use expired, moldy, damaged, or otherwise abnormal-appearing medicine, and immediately notify the manufacturer.
Expiry date: 24 months from the manufacturing date.
Storage: In a dry place, at a temperature not exceeding 30°C, away from sunlight.
Packaging specifications: Box of 4 blisters x 5 lozenges; Box of 20 blisters x 5 lozenges
Usage standard: TCCS
Registration number: TCT-00114-23
Manufacturing plant meets GMP-WHO, GLP, GSP standards
Responsible for the product & Distributed by: HOA LINH PHARMACEUTICAL JOINT STOCK COMPANY
Address: 56 Truong Cong Giai, Dich Vong Ward, Cau Giay District, Hanoi City.
Manufacturing plant: Lot B3 Phung Industrial Park, Dan Phuong District, Hanoi City.
Can't find the product you're looking for??
Share your needs with suppliers and receive direct quotes using Arobid RFQ!